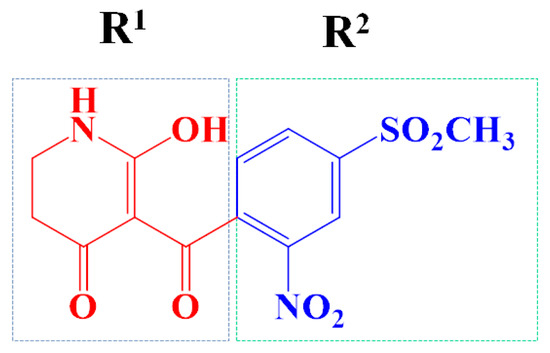
IJMS | Free Full-Text | Computer-Aided and AILDE Approaches to Design Novel 4-Hydroxyphenylpyruvate Dioxygenase Inhibitors
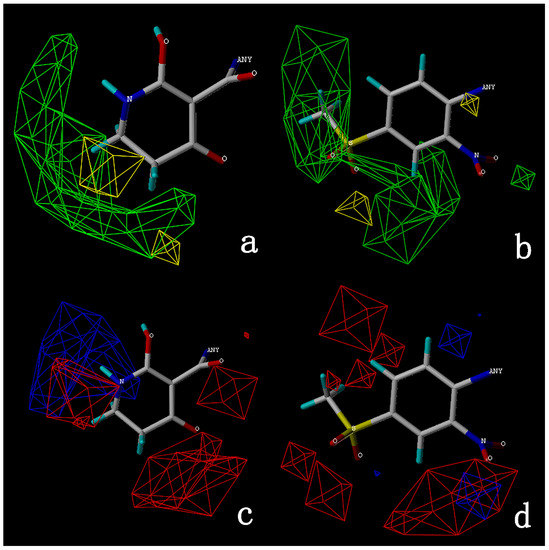
IJMS | Free Full-Text | Computer-Aided and AILDE Approaches to Design Novel 4-Hydroxyphenylpyruvate Dioxygenase Inhibitors
THEMIS-SHP1 Recruitment by 4-1BB Tunes LCK-Mediated Priming of Chimeric Antigen Receptor-Redirected T Cells

QSAR study of unsymmetrical aromatic disulfides as potent avian SARS-CoV main protease inhibitors using quantum chemical descriptors and statistical methods - ScienceDirect

IJMS | Free Full-Text | Computer-Aided and AILDE Approaches to Design Novel 4-Hydroxyphenylpyruvate Dioxygenase Inhibitors

PDF) Predictions of Cleavability of Calpain Proteolysis by Quantitative Structure-Activity Relationship Analysis Using Newly Determined Cleavage Sites and Catalytic Efficiencies of an Oligopeptide Array
![Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2- -trifluoroethyl)pyrrolidine-1-carb oxamide and solid state forms thereof Patent Grant Pangan , et al. May 4, 2 [AbbVie Inc.] Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2- -trifluoroethyl)pyrrolidine-1-carb oxamide and solid state forms thereof Patent Grant Pangan , et al. May 4, 2 [AbbVie Inc.]](https://uspto.report/patent/grant/10995095/US10995095-20210504-C00002.png)
Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2- -trifluoroethyl)pyrrolidine-1-carb oxamide and solid state forms thereof Patent Grant Pangan , et al. May 4, 2 [AbbVie Inc.]
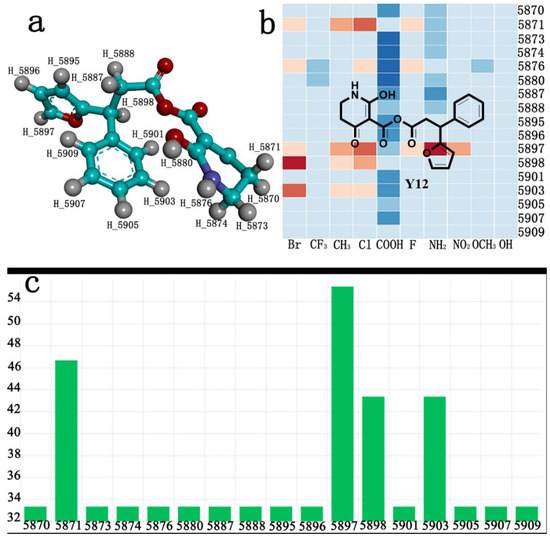
IJMS | Free Full-Text | Computer-Aided and AILDE Approaches to Design Novel 4-Hydroxyphenylpyruvate Dioxygenase Inhibitors
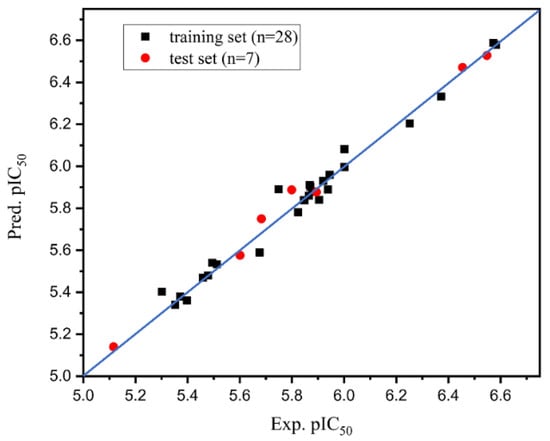
IJMS | Free Full-Text | Computer-Aided and AILDE Approaches to Design Novel 4-Hydroxyphenylpyruvate Dioxygenase Inhibitors
THEMIS-SHP1 Recruitment by 4-1BB Tunes LCK-Mediated Priming of Chimeric Antigen Receptor-Redirected T Cells

Total Synthesis and Biological Activity of Marine Alkaloid Eudistomins Y1–Y7 and Their Analogues – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open







